Team:Dalton School NY/Notebook
From 2012hs.igem.org
(→BsaI-digested Miniprep DNA) |
|||
| Line 16: | Line 16: | ||
[[Image:Dalton_Fig8.jpg]] | [[Image:Dalton_Fig8.jpg]] | ||
| + | |||
| + | Based on the data above, we concluded that the following clones had inserts of the correct size: | ||
| + | |||
| + | '''Promoters''' | ||
| + | |||
| + | MET15 yes: B | ||
| + | |||
| + | GAL1 yes: A | ||
| + | |||
| + | GAL1-L yes: A | ||
| + | |||
| + | GAL1-S yes: B | ||
| + | |||
| + | RAD52 yes: B | ||
| + | |||
| + | RAD50 yes: A | ||
| + | |||
| + | RAD54 yes: A, B | ||
| + | |||
| + | RAD55 yes: A, B | ||
| + | |||
| + | MRE11 yes: A, B | ||
| + | |||
| + | XRS2 yes: A, B | ||
| + | |||
| + | REV1 yes: B | ||
| + | |||
| + | TDH3 yes: A, B | ||
| + | |||
| + | LIF1 '''none''' | ||
| + | |||
| + | MIG1 yes: B | ||
| + | |||
| + | TUB1 yes: A | ||
| + | |||
| + | CSE4 yes: B | ||
| + | |||
| + | CLN2 yes: B | ||
| + | |||
| + | CLB2 '''none''' | ||
| + | |||
| + | ACT1 '''none''' | ||
| + | |||
| + | ENO1 '''none''' | ||
| + | |||
| + | ENO2 '''none''' | ||
| + | |||
| + | TEF2 yes: B | ||
| + | |||
| + | TEF1 '''none''' | ||
| + | |||
| + | EAF6 '''none''' | ||
| + | |||
| + | HSP12 yes: A, B | ||
| + | |||
| + | HSP26 yes: A | ||
| + | |||
| + | HSC82 '''none''' | ||
| + | |||
| + | MSN2 yes: A | ||
| + | |||
| + | MSN4 '''none''' | ||
| + | |||
| + | HHO1 yes: A, D | ||
| + | |||
| + | |||
| + | '''Protein-coding sequences''' | ||
| + | |||
| + | BFP maybe: A (the insert may be too large) | ||
| + | |||
| + | GFP '''none''' | ||
| + | |||
| + | YFP '''none''' | ||
| + | |||
| + | mTangerine '''none''' | ||
| + | |||
| + | mCherry yes: A (but bottom band seems possibly too intense) | ||
| + | |||
| + | mGrape1 '''none''' | ||
Return to our [https://2012hs.igem.org/Team:Dalton_School_NY main page]. | Return to our [https://2012hs.igem.org/Team:Dalton_School_NY main page]. | ||
Revision as of 02:40, 1 June 2012
In progress...
Testing of DNA prepared by several methods
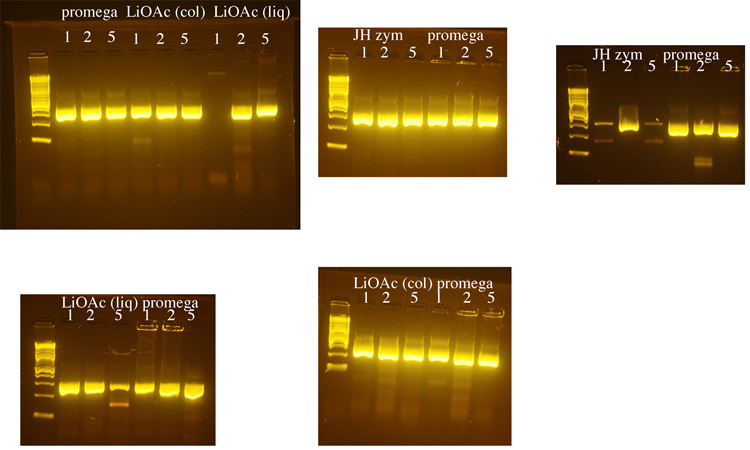
Yeast genomic DNA is commonly prepared using a phenol/chloroform based method. Because both phenol and chloroform are toxic chemicals, we tested several methods for preparing yeast genomic DNA that do not use these chemicals. We compared a method that uses LiOAc to disrupt the yeast cell wall (BioTechniques 50:325-328, 2011) to methods that use the enzyme zymolyase to disrupt the cell wall (a Promega kit or a protocol from Jim Haber’s lab, “JH zym”). The Haber lab protocol produced the greatest yield of genomic DNA, but DNA made using the Promega kit was used in subsequent PCRs because it seemed to produce the most consistent results. 1µl, 2µl, or 5µl of genomic DNA was used in each PCR reaction with LIF1 promoter primers. Following these initial tests, 1µl of yeast genomic DNA was used for subsequent PCRs. The LiOAc method was the shortest and least expensive and we may switch to this method when future DNA preps are needed.
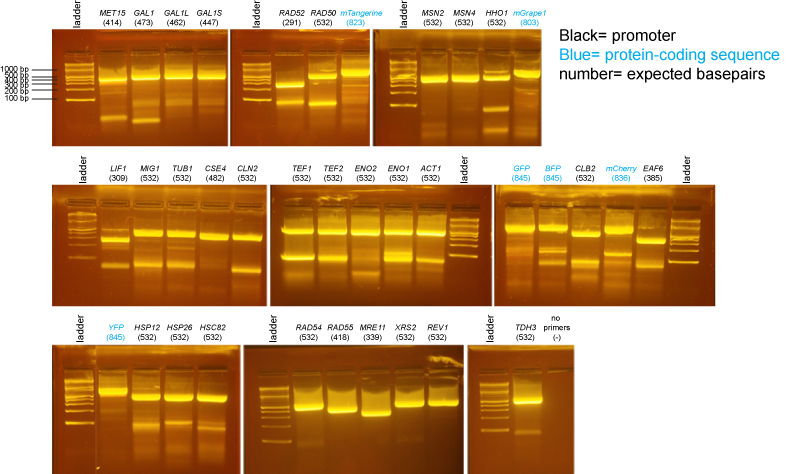
PCR of promoters and fluorescent protein coding sequences
BsaI-digested Miniprep DNA
The samples on these gels contain BsaI-digested miniprep DNA prepared from individual colonies from the transformations above.
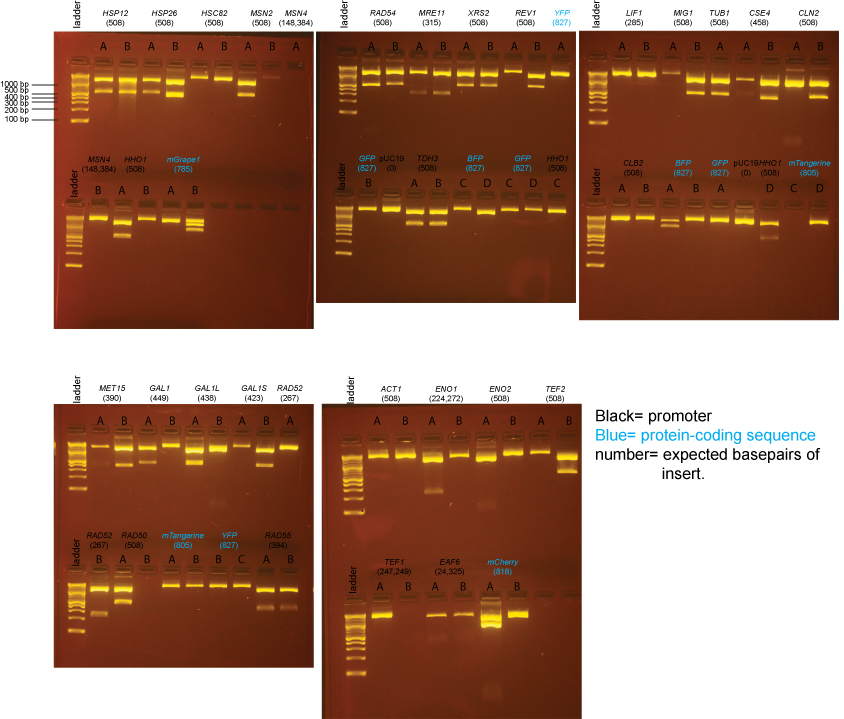
Based on the data above, we concluded that the following clones had inserts of the correct size:
Promoters
MET15 yes: B
GAL1 yes: A
GAL1-L yes: A
GAL1-S yes: B
RAD52 yes: B
RAD50 yes: A
RAD54 yes: A, B
RAD55 yes: A, B
MRE11 yes: A, B
XRS2 yes: A, B
REV1 yes: B
TDH3 yes: A, B
LIF1 none
MIG1 yes: B
TUB1 yes: A
CSE4 yes: B
CLN2 yes: B
CLB2 none
ACT1 none
ENO1 none
ENO2 none
TEF2 yes: B
TEF1 none
EAF6 none
HSP12 yes: A, B
HSP26 yes: A
HSC82 none
MSN2 yes: A
MSN4 none
HHO1 yes: A, D
Protein-coding sequences
BFP maybe: A (the insert may be too large)
GFP none
YFP none
mTangerine none
mCherry yes: A (but bottom band seems possibly too intense)
mGrape1 none
Return to our main page.
 "
"